
Missouri University of Science and Technology. "Infinite Polyiodide Chains in the Pyrroloperylene-Iodine Complex: Insights into the Starch–Iodine and Perylene–Iodine Complexes". Iodine is often used to test foods for starch. The new spacings absorb visible light differently and create the deep blue color. This causes a change in the arrangement of electrons and energy level spacings. Brilliant Biology Student Master Biology Labs. The starch pushes the iodine into a line in the middle of the amylose coils and creates a transfer of charge between the iodine and starch. "Ein sehr empfindliches Reagens für Jodine, aufgefunden in der Stärke (Amidon)". "Mémoire sur les Combinaisons de l'Iode avec les Substances Végétales et Animales". "Kinetic Analysis of Amylase Using Quantitative Benedict's and Iodine Starch Reagents".
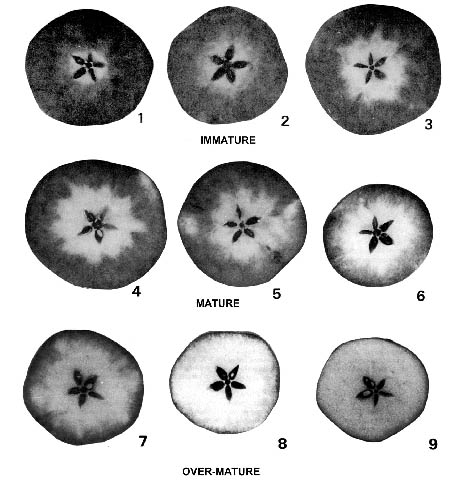
^ a b Cochran, Beverly Lunday, Deborah Miskevich, Frank (2008).The color change can be used to detect moisture or perspiration, as in the Minor test or starch–iodine test. Put a few drops of the iodine solution on each one of the food items. It showed a negative result which reflected that reducing sugar was absent in green bean. Since the yellow-orange iodine solution stayed the same color when it was put on the. The starch index is an arbitrary number that corresponds with a certain amount of starch loss. When a cross-section of an apple is sprayed with an iodine solution, a characteristic pattern of stained and unstained flesh occurs. Close to the endpoint, the starch is added, and the titration process is resumed taking into account the amount of thiosulfate added before adding the starch. If iodine solution reacts with starch, it will change color into black. Areas of the fruit that have lost starch will remain unstained. This is due to the insolubility of the starch–triiodide complex which may prevent some of the iodine reacting with the titrant. During iodine titrations, concentrated iodine solutions must be reacted with some titrant, often thiosulfate, in order to remove most of the iodine before the starch is added. The colour of the starch complex is so deep, that it can be detected visually when the concentration of the iodine is as low as 20 µM at 20 ☌. However, the complex is not formed if only iodine or only iodide (I −) is present. Starch forms a very dark blue-black complex with triiodide. In this test, iodine and starch form a distinct blue-black colored complex. It also helps to distinguish between mono or disaccharides from polysaccharides (glycogen, dextrin, and amylase). This test helps to identify the presence of starch in a sample. Starch is often used in chemistry as an indicator for redox titrations where triiodide is present. The iodine test is a chemical reaction-based identification test for starch.
